| Lysozyme navigation: | Home | Copyright | Contact us | |
|
Enzyme
Sequence
Crystallization
Structure
Bibliography
anti-HIV
Lysozyme
published: 10 Oct 2006 (22:04)
Lysozyme is 129 aminoacid residues enzyme (EC 3.2.1.17), hydrolase which catalyzes hydrolysis of 1,4-beta-linkages between N-acetylmuramic acid and N-acetyl-D-glucosamine residues in peptidoglycan and between N-acetyl-D-glucosamine residues in chitodextrins. Molecular weight of Lysozyme is an approximately 14.7 kDa. Alternative name for Lysozyme are 1,4-N-acetylmuramidase, L-7001, N,O-diacetylmuramidase, PR1-Lysozyme, Globulin G1, Globulin G, Lysozyme G, Mucopeptide N-acetylmuramoylhydrolase, Mucopeptide glucohydrolase and Muramidase. This catalytic activity is non-specifically targeted to the bacterial cell membranes and related with general non-specific organism defence.Lysozyme is present in the mucosal secretion such as saliva and tears. In high concentration, about 3% from all proteins, Lysozyme is present in chicken egg-white. This enzyme is only effective against Gram positive bacterial cells. Gram negative bacteria and yeast are completely resistant to lysing by it.
Historically, Lysozyme was discovered in 1922 by Alexander Fleming (Fleming A. (1922) On a remarkable becteriolytic element found in tissues and secretion. Proc Roy Soc Ser B, 93, 306-317.). This enzyme was discovered by accident, which was happen in the Fleming's lab. The nasal drippings were accidentally occurring in the petre dish with bacterial culture and these cells were lysed. This phenomenon was carefully investigated and the main acting enzyme was identified as Lysozyme.
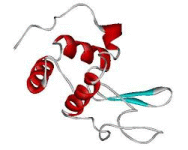
In 1965 the structure of Lysozyme was solved by X-Ray analysis with 2 angstrom resolution by David Chilton Phillips. For many years Lysozyme was the best object for X-Ray analysis due to many unique properties of this enzyme. First of all Lysozyme is easy to purify from egg-white. Secondly, this protein is very easy to crystallize, which is not the case for most of the other proteins. This feature of Lysozyme is widely used for it purification. And finally, crystals of Lysozyme diffract X-Ray beam to a very high resolution, currently the highest resolution structure, presented in Protein Data Bank, was solved at resolution 0.94 Angstrom.
In viruses (or bacteriophages), Lysozyme is used as an agent to break into the host bacterial cell. Lysozyme from the tail of the virus (or bacteriophage) destroys the peptidoglycan bacterial cell wall and then virus can injects its DNA. After multiplication in bacteria, many Lysozyme molecules are created to lyse the bacterial cell wall and release new viruses.
News
Lysozome deficient cell has better immunity against SalmonellaBy replacing one of the most important soldiers ...
Lysozyme Decrease Diseases Caused by SalmonellaWhen a hen lays an egg, it gives the egg some ...
Other news pages:
1
| Copyright 2006-... by Lysozyme.co.uk |